World Gout Day | Hydrogen, Light, Infrared: Three New Approaches for Gout Management
2026-04-21
April 20th of each year is "World Gout Day". The selection of this date holds special significance - the diagnostic threshold for hyperuricemia in adult men is exactly 420 μmol/L. When the blood uric acid level exceeds this limit, urate crystals may gradually deposit in the joints, and suddenly trigger severe redness, swelling, heat and pain during a big meal or after drinking alcohol.
Gout is essentially a metabolic disorder, often occurring concurrently with insulin resistance, obesity, hypertension, and abnormal blood lipids. Besides standard uric acid-lowering drug therapy and strict dietary control, a series of non-drug adjunctive therapies have gradually come into the research spotlight in recent years.
Hydrotherapy: From Antioxidation to Reducing Uric Acid
Hydrotherapy (inhalation of hydrogen or consumption of hydrogen-rich water) is one of the most highly evidenced complementary therapies in the field of gout.
A high-quality randomized double-blind controlled trial conducted in 2024 showed that after 8 weeks of continuous consumption of high-concentration hydrogen-rich water by patients with hyperuricemia, the blood uric acid level significantly decreased from the baseline of 488.2 μmol/L to 446.8 μmol/L. Further animal experiments revealed the mechanism: hydrogen-rich water can selectively eliminate harmful free radicals and significantly reduce the levels of key inflammatory factors TNF-α and IL-1β in the bodies of gouty rats, thereby alleviating joint swelling.
The unique advantage of hydrogen molecules lies in their small size, which enables them to easily penetrate the cell membrane and reach the mitochondria. They can precisely "extinguish" excessive oxidative stress while protecting the normal functions of cells - this is precisely the core driver of the local inflammatory storm in the joints during a gout attack.
Infrared therapy: Improving circulation, reviving cellular metabolism
When it comes to infrared rays, many people's first thought is "heat therapy", but recent research has shown that the effects of far-infrared rays go far beyond the thermal effect.
Studies have shown that far-infrared radiation can activate the AMPK signaling pathway within cells - this pathway is the "master switch" for cellular energy metabolism. In a diabetic mouse model, 30-minute daily exposure to far-infrared radiation for two weeks significantly reduced fasting blood sugar and protected pancreatic β cells from apoptosis. For patients with gout, the activation of this pathway means better insulin sensitivity and a more stable metabolic environment, and insulin resistance is a common "accomplice" of hyperuricemia.
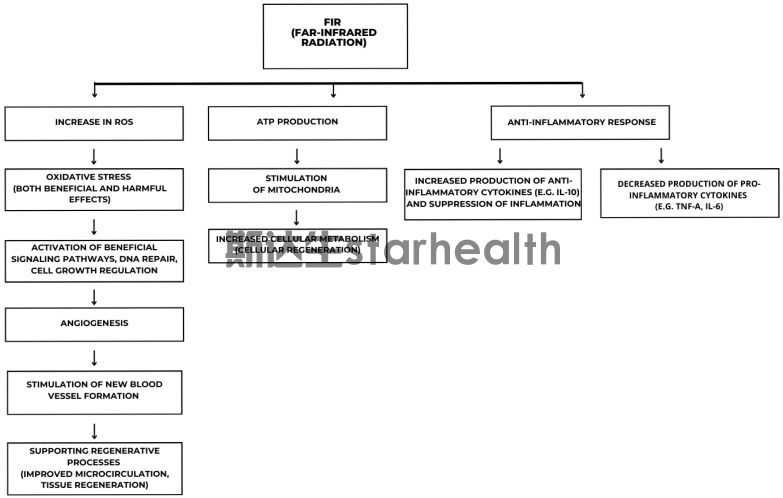
What's more interesting is that a small clinical study on patients with abnormal blood lipids found that 15-minute sessions of far-infrared radiation (with a wavelength of 9-12 μm) once a week resulted in an average decrease of approximately 13% in LDL cholesterol levels after 3 weeks. The triglyceride levels also dropped significantly from 309 mg/dL to 155 mg/dL. At the same time, the blood flow velocity in the retinal vessels significantly increased, suggesting that far-infrared radiation can effectively improve microcirculation - this is undoubtedly good news for the local blood supply of joints damaged by urate crystals.
Safety reminder: When exposed to far-infrared rays, be sure to wear protective eye goggles; the recommended duration of each exposure is 15 to 30 minutes, and the skin surface temperature should not exceed 42℃; patients with implanted electronic devices such as cardiac pacemakers need to carefully assess the situation.
Red light/near-infrared light: Charging mitochondria
At the junction of visible light and infrared light, red light with a wavelength of approximately 670 nm has attracted much attention in recent years.
A 2024 study conducted on healthy volunteers found that a mere 15-minute exposure to 670 nm red light could reduce the area under the blood glucose curve by 7.9% and decrease the increase in blood glucose by 27.7% in a subsequent oral glucose tolerance test. The researchers believe that this is directly related to the activation of cytochrome c oxidase on the mitochondrial respiratory chain by red light, the increase in ATP production, and the acceleration of cells' uptake of glucose.
Although there are currently no large-scale red light studies specifically targeting patients with gout, considering the close correlation between gout and high blood sugar as well as insulin resistance, and the anti-inflammatory and analgesic effects of red light that have been demonstrated in animal models of arthritis, this "charging cells" approach is worthy of continued attention.
Three paths, one common target: Mitochondria and Inflammation
Whether it's hydrogen molecules, far-infrared rays, or red light, their mechanisms of action ultimately all point to the same cellular organelle - the mitochondria.
Mitochondria are not only the "power plants" of cells, but also the main source of reactive oxygen species. When metabolic disorders occur, mitochondrial function declines, and excessive reactive oxygen species are produced, leading to oxidative stress and chronic low-grade inflammation - this is precisely the common ground for metabolic diseases such as gout, diabetes, and fatty liver.
Hydrogen molecules selectively neutralize harmful free radicals. Far-infrared rays activate AMPK to enhance mitochondrial function. Red light directly stimulates the mitochondrial respiratory chain to increase energy output - these three pathways "repair" the same damaged system from different angles. This is precisely the fundamental reason why they are highly anticipated.
It is important to note that the aforementioned therapy is currently classified as an adjunctive treatment. Most of the evidence for far-infrared and phototherapy comes from preclinical models and small-scale trials. Different studies vary greatly in parameters such as wavelength, irradiation duration, and frequency, and there is still a long way to go before a standardized clinical guideline can be formed.
For patients with gout, regular use of uric acid-lowering drugs and maintaining blood uric acid levels within the target range (generally < 360 μmol/L, and < 300 μmol/L for those with tophi) remains the cornerstone for preventing joint damage and kidney injury. Hydrogen, light, and far-infrared non-drug therapies are more suitable as "add-ons" rather than replacements in the comprehensive management plan.
Reference materials
Nowacka A, et al. Therapeutic Potential of Infrared and Related Light Therapies in Metabolic Diseases. Int J Mol Sci. 2025;26(11):5134.
*Hsu YH, et al. Far-infrared radiation prevents decline in β-cell mass and function in diabetic mice. Metabolism. 2020;104:154143.*
Powner MB, Jeffery G. Light stimulation of mitochondria reduces blood glucose levels. J Biophotonics. 2024;17(3):e202300521.
Fenglin Wu , et al. Effects of hydrogen-rich water on blood uric acid in patients with hyperuricemia: A randomized placebo-controlled trial.Heliyon. 2024 Aug 15;10(16):e36401.




